You will not be allowed to compare more than 4 products at a time
View compareReturns
Medsitis offers a 30 day return policy. The 30 days begin on the date of delivery. Return requests must be initiated by contacting customer service at cs@medsitis.com. If the return is initiated due to no fault of Medsitis, customer is responsible for the return freight. Medsitis charges a reasonable restock fees to cover returns operations where it is appropriate. For more information view our full return policy here
Domestic Shipping (48 Contiguous States | Excludes Alaska & Hawaii):
Standard Shipping (1-10 Business Days):
Order Under $79 - $6.99
Order Over $79 - Free Shipping
Expedited Shipping (1-5 Business Days):
Order Under $79 - $9.99
Order Over $79 - $7.99
International Shipping (Outside 48 contiguous states):
Real-Time shipping is calculated at checkout and is based on your shipping service selection.
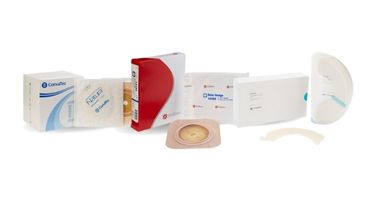
Aquacel® AG Non-Adhesive Foam Dressing with Silver - Sterile
The only silver foam dressing that offers the healing benefits of an Aquacel® AG interface layer, the comfort of foam and the bacteria-killing power of ionic silver.*1
Waterproof/bacteria barrier provides:
- Controlled moisture vapor transmission*
- Protection against viral/bacterial penetration*2
Soft foam pad absorbs fluid*
AQUACEL® AG interface with Hydrofiber® Technology gels on contact with the wound surface to:
- Lock in wound exudate, including harmful components*3
- Help maintain a moist wound healing environment
- Help prevent lateral spread of fluid, reducing risk of maceration*8
- Micro-contour to the wound bed, minimizing dead space where bacteria can grow*9
- Kill a wide variety of micro-organisms, including certain tested antibiotic-resistant superbugs, with powerful ionic silver*1
*As demonstrated in vitro
Item # | Size:
420639 | 2" x 2" (5 cm x 5 cm)
420642 | 4" x 4" (10 cm x 10 cm)
420645 | 6" x 6" (15 cm x 15 cm)
420646 | 8" x 8" (20 cm x 20 cm)
420806 | 6" x 8" (15 cm x 20 cm)
References:
1. Jones SA, Bowler PG, Walker M, Parsons D. Controlling wound bioburden with a novel silver-containing Hydrofiber dressing. Wound Repair Regen. 2004;12(3):288-294.
2. AQUACEL® Foam dressing—waterproofness, bacterial and viral barrier testing. WHR13538 MS069. Data on File, ConvaTec Inc.
3. Walker M, Hobot JA, Newman GR, Bowler PG. Scanning electron microscopic examination of bacterial immobilization in a carboxymethyl cellulose (AQUACEL®) and alginate dressings. Biomaterials. 2003;24(5):883-890.
4. Armstrong SH, Brown DA, Hill E, Ruckley CV. A randomized trial of a new Hydrofiber dressing, AQUACEL®, and an alginate in the treatment of exuding leg ulcers. Presented at: 5th European Conference on Advances in Wound Management; Harrogate, UK: November 1995.
5. Barnea Y, Amir A, Leshem D, et al. Clinical comparative study of AQUACEL® and paraffin gauze dressing for split skin donor site treatment. Ann Plast Surg. 2004;53(2):132-136.
6. Caruso DM, Foster KN, Blome Eberwein SA, et al. Randomized clinical study of Hydrofiber dressing with silver or silver sulfadiazine in the management of partial thickness burns. J Burn Care Res. 2006;27(3):298-309.
7. Kogan L, Moldavsky M, Szvalb S, Govrin Yehudain J. Comparative study of AQUACEL® and Silverol treatment in burns. Ann Burns Fire Disasters. 2004;17(4):201-207.
8. Waring MJ, Parsons D. Physico-chemical characterisation of carboxymethylated spun cellulose fibres. Biomaterials. 2001;22(9):903-912.
9. Jones SA, Bowler PG, Walker M. Antimicrobial activity of silver-containing dressings is influenced by dressing conformability with a wound surface. WOUNDS. 2005;17(9):263-270.
(c) 2013 ConvaTec Inc.
(R)/TM AQUACEL and Hydrofiber are trademarks of ConvaTec Inc. All other trademarks are the property of their respective owners.
Returns Policy
You may return most new, unopened items within 30 days of delivery for a full refund. We'll also pay the return shipping costs if the return is a result of our error (you received an incorrect or defective item, etc.).
You should expect to receive your refund within four weeks of giving your package to the return shipper, however, in many cases you will receive a refund more quickly. This time period includes the transit time for us to receive your return from the shipper (5 to 10 business days), the time it takes us to process your return once we receive it (3 to 5 business days), and the time it takes your bank to process our refund request (5 to 10 business days).
If you need to return an item, simply login to your account, view the order using the "Complete Orders" link under the My Account menu and click the Return Item(s) button. We'll notify you via e-mail of your refund once we've received and processed the returned item.
Shipping
We can ship to virtually any address in the world. Note that there are restrictions on some products, and some products cannot be shipped to international destinations.
When you place an order, we will estimate shipping and delivery dates for you based on the availability of your items and the shipping options you choose. Depending on the shipping provider you choose, shipping date estimates may appear on the shipping quotes page.
Please also note that the shipping rates for many items we sell are weight-based. The weight of any such item can be found on its detail page. To reflect the policies of the shipping companies we use, all weights will be rounded up to the next full pound.